Chlor Alkali Process












Chlor Alkali Process
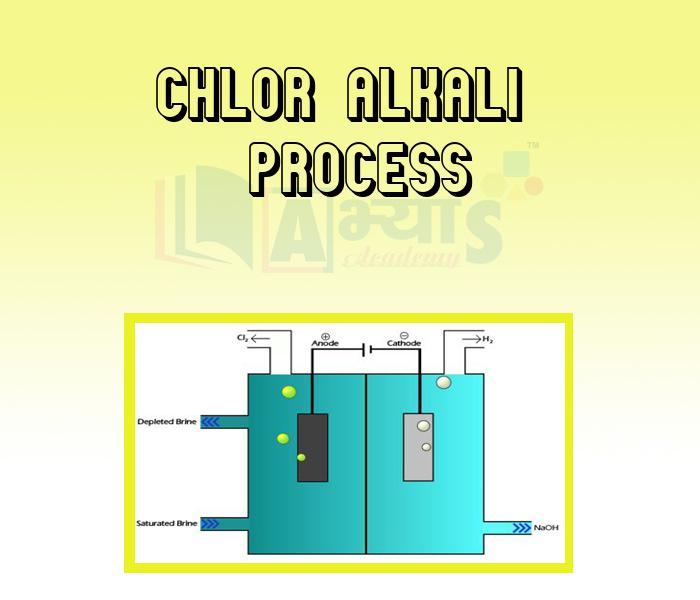
Chlor -alkali process: When electricity is passed through an aqueous solution of sodium chloride, it decomposes to form sodium hydroxide, chlorine gas and hydrogen gas.
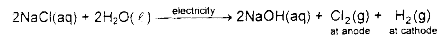
• When electricity is passed through the solution of sodium chloride, it splits up into sodium ions and chloride ion.
• Sodium ion combines with the hydroxyl ion of water to form sodium hydroxide.
• Chloride ions lose electron at anode and form chlorine atom.
• Two chlorine atoms combine to form a chlorine molecule.
• H of water gains electron at cathode and change into hydrogen atom.
When electricity is passed through an aqueous solution of sodium chloride, it decomposes to form ________________________. | |||
| Right Option : D | |||
| View Explanation | |||
The aqueous solution of this substance will not conduct electric current ? | |||
| Right Option : C | |||
| View Explanation | |||
Which of the following gases is at the anode in chlor alkali process? | |||
| Right Option : B | |||
| View Explanation | |||
Students / Parents Reviews [10]
It was a good experience with Abhyas Academy. I even faced problems in starting but slowly and steadily overcomed. Especially reasoning classes helped me a lot.

Cheshta
10thA marvelous experience with Abhyas. I am glad to share that my ward has achieved more than enough at the Ambala ABHYAS centre. Years have passed on and more and more he has gained. May the centre flourish and develop day by day by the grace of God.

Archit Segal
7thIt has a great methodology. Students here can get analysis to their test quickly.We can learn easily through PPTs and the testing methods are good. We know that where we have to practice

Barkha Arora
10thMy experience with Abhyas is very good. I have learnt many things here like vedic maths and reasoning also. Teachers here first take our doubts and then there are assignments to verify our weak points.

Shivam Rana
7thIt was good as the experience because as we had come here we had been improved in a such envirnment created here.Extra is taught which is beneficial for future.

Eshan Arora
8thAbhyas Methodology is very good. It is based on according to student and each child manages accordingly to its properly. Methodology has improved the abilities of students to shine them in future.

Manish Kumar
10thMy experience was very good with Abhyas academy. I am studying here from 6th class and I am satisfied by its results in my life. I improved a lot here ahead of school syllabus.

Ayan Ghosh
8thOne of the best institutes to develope a child interest in studies.Provides SST and English knowledge also unlike other institutes. Teachers are co operative and friendly online tests andPPT develope practical knowledge also.

Aman Kumar Shrivastava
10thBeing a parent, I saw my daughter improvement in her studies by seeing a good result in all day to day compititive exam TMO, NSO, IEO etc and as well as studies. I have got a fruitful result from my daughter.

Prisha Gupta
8thAbout Abhyas metholodology the teachers are very nice and hardworking toward students.The Centre Head Mrs Anu Sethi is also a brilliant teacher.Abhyas has taught me how to overcome problems and has always taken my doubts and suppoeted me.
